PERFORMANCE OF A MICROFLUIDIC POINT-OF-CARE IMMUNOASSAY FOR D-DIMER COMPARED WITH THE SYSMEX CS-2500 ANALYZER IN THE EXCLUSION OF VENOUS THROMBOEMBOLISM: A COMPARATIVE ANALYSIS
DOI:
https://doi.org/10.55374/jseamed.v10.289Keywords:
D-dimer, point-of-care testing, venous thromboembolism, diagnostic accuracy, microfluidicsAbstract
Background: D-dimer testing is central to venous thromboembolism (VTE) exclusion pathways, but reliance on central laboratory analyzers may delay downstream diagnostic decisions, prolong time to imaging, and defer safe exclusion in patients with low or intermediate clinical pre-test probability.
Objectives: To evaluate the analytical agreement and clinical diagnostic performance of a novel microfluidic point-of-care D-dimer assay (mLabs®) against the Sysmex CS-2500 central laboratory analyzer.
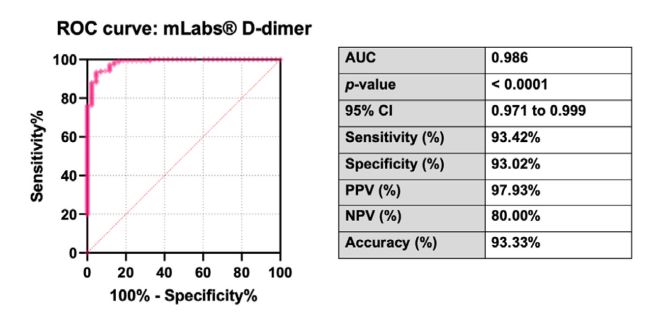
Methods: In this prospective single-center method-comparison study, 195 paired patient specimens were analyzed using the mLabs® device with 250 µL whole blood and the Sysmex CS-2500 analyzer with 50 µL citrated plasma. For threshold-based categorical comparison, the Sysmex CS-2500 served as the reference method at the 500 ng/mL FEU cutoff. Correlation, agreement, and diagnostic accuracy were assessed using Pearson correlation, intraclass correlation coefficient (ICC), Bland–Altman analysis, and receiver operating characteristic (ROC) analysis, including sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV).
Results: The mLabs® assay correlated strongly with the reference method (r = 0.94; ICC = 0.96). Sensitivity and specificity at the 500 ng/mL FEU cutoff were 93.4% and 93.0%, respectively; PPV was 97.9%, and NPV was 80.0%; and the area under the ROC curve was 0.986. However, Bland–Altman analysis demonstrated a mean negative bias of −483.5 ng/mL, and 10 Sysmex reference-positive samples were classified as negative by mLabs®. In contrast, 3 Sysmex reference-negative samples were classified as positive.
Conclusion: Although the microfluidic point-of-care D-dimer assay (mLabs®) showed strong analytical agreement and excellent overall discrimination, the observed discordant mLabs®-negative/Sysmex-positive results and negative predictive value of 80.0% relative to the reference method indicate that mLabs® should not be assumed to be interchangeable with Sysmex CS-2500 at the 500 ng/mL FEU rule-out threshold. Its most appropriate role is within structured diagnostic algorithms that incorporate clinical pre-test probability.
Downloads
References
Anderson Jr FA, Spencer FA. Risk factors for venous thromboembolism. Circulation 2003; 107(23_suppl_1): I–9. DOI: https://doi.org/10.1161/01.CIR.0000078469.07362.E6
Bates SM, Jaeschke R, Stevens SM, Goodacre S, Wells PS, Stevenson MD, et al. Diagnosis of DVT: antithrombotic therapy and prevention of thrombosis: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2012; 141(2): e351S-418S. DOI: https://doi.org/10.1378/chest.141.4.1129
Konstantinides SV, Meyer G, Becattini C, Bueno H, Geersing GJ, Harjola VP, et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism developed in collaboration with the European Respiratory Society (ERS), The Task Force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC). Eur Heart J 2020; 41: 543–603. DOI: https://doi.org/10.1093/eurheartj/ehz405
Di Nisio M, Squizzato A, Rutjes AW, Büller HR, Zwinderman AH, Bossuyt PM. Diagnostic accuracy of D‐dimer test for exclusion of venous thromboembolism: a systematic review. J Thromb Haemost 2007; 5: 296–304. DOI: https://doi.org/10.1111/j.1538-7836.2007.02328.x
Perveen S, Unwin D, Shetty AL. Point of care D-dimer testing in the emergency department: a bioequivalence study. Ann Lab Med 2013; 33: 34–8. DOI: https://doi.org/10.3343/alm.2013.33.1.34
Price CP, Fay M, Hopstaken RM. Point-ofcare testing for d-dimer in the diagnosis of venous thromboembolism in primary care: A narrative review. Cardiol Ther 2021; 10: 27–40. DOI: https://doi.org/10.1007/s40119-020-00206-2
Heerink JS, Oudega R, Gemen E, Hopstaken R, Koffijberg H, Kusters R. Are the latest point-of-care D-dimer devices ready for use in general practice? A prospective clinical evaluation of five test systems with a capillary blood feature for suspected venous thromboembolism. Thromb Res 2023; 232: 113–22. DOI: https://doi.org/10.1016/j.thromres.2023.10.014
Elf J, Strandberg K, Svensson P. Performance of two relatively new quantitative D-dimer assays (Innovance D-dimer and AxSYM D-dimer) for the exclusion of deep vein thrombosis. Thromb Res 2009; 124: 701–5. DOI: https://doi.org/10.1016/j.thromres.2009.07.008
Buderer NMF. Statistical methodology: I. Incorporating the prevalence of disease into the sample size calculation for sensitivity and specificity. Acad Emerg Med 1996; 3: 895– 900. DOI: https://doi.org/10.1111/j.1553-2712.1996.tb03538.x
Negida A, Fahim NK, Negida Y. Sample size calculation guide-part 4: how to calculate the sample size for a diagnostic test accuracy study based on sensitivity, specificity, and the Area under the ROC curve. Adv J Emerg Med 2019; 3: e33.
Arunothai S, Panpikoon T, Boonyawat K, Sutherasan Y, Theerawit P. Prevalence and incidence of deep vein thrombosis in medical critically ill patients: a prospective analysis in a single tertiary care centre in Thailand. Presented at: International Society on Thrombosis and Haemostasis; 2021. Philadelphia, USA.
Bland JM, Altman D. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986; 327: 307–10. DOI: https://doi.org/10.1016/S0140-6736(86)90837-8
Rostami M, Mansouritorghabeh H. D-dimer level in COVID-19 infection: a systematic review. Expert Rev Hematol 2020; 13: 1265– 75. DOI: https://doi.org/10.1080/17474086.2020.1831383
Borowiec A, Dąbrowski R, Kowalik I, Rusinowicz T, Hadzik-Błaszczyk M, Krupa R, et al. Elevated levels of d-dimer are associated with inflammation and disease activity rather than risk of venous thromboembolism in patients with granulomatosis with polyangiitis in long term observation. Adv Med Sci 2020; 65: 97–101. DOI: https://doi.org/10.1016/j.advms.2019.12.007
Righini M, Van Es J, Den Exter PL, Roy PM, Verschuren F, Ghuysen A, et al. Age-adjusted D-dimer cutoff levels to rule out pulmonary embolism: the ADJUST-PE study. JAMA 2014; 311: 1117–24. DOI: https://doi.org/10.1001/jama.2014.2135
Reber G, Bounameaux H, Perrier A, De Moerloose P. A new rapid point-of-care D-dimer enzyme-linked immunosorbent assay (Stratus CS D-dimer) for the exclusion of venous thromboembolism. Blood Coagul Fibrinolysis 2004; 15: 435–8. DOI: https://doi.org/10.1097/01.mbc.0000114443.59147.c4
Fukuda T, Kasai H, Kusano T, Shimazu C, Kawasugi K, Miyazawa Y. A rapid and quantitative D-Dimer assay in whole blood and plasma on the point-of-care PATHFAST analyzer. Thromb Res 2007; 120: 695–701. DOI: https://doi.org/10.1016/j.thromres.2006.12.021
Chantkran W, Jamnarnwej P, Sritanabutr P, Arnutti P. Evaluation of point‐of‐care testing device for anemia detection: A cross‐sectional method comparison study from Thailand. J Clin Lab Anal 2021; 35: e23976. DOI: https://doi.org/10.1002/jcla.23976
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Journal of Southeast Asian Medical Research

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
The Journal of Southeast Asian Medical Research will hold the copyright to all published articles. The publisher's production department handles copyright forms once a manuscript is accepted and scheduled for publication.







